By Professor Emeritus, Paul D Blumenthal, Stanford University School of Medicine, Department of Obstetrics and Gynecology
Twenty-five years ago I was honored to be part of the development of VIA as the screening component for cervical cancer prevention programs. However, despite improvements in a variety of screening methodologies, the rates of cervical cancer have increased by 45% in the last 30 years.

In recent years there has been a shift to recognize that defeating cervical cancer has three essential components: the screening coverage, the treatment effectiveness, and the link between screening and treatment. Screening by itself has no intrinsic preventative value no matter how accurate the test, therefore to be effective, tests must be linked to treatment, and the closer the temporal link, the better.
In this article, I’m going to share some of the latest innovations in cervical cancer screening and treatment as we work towards eradicating this preventable disease.
Advances in screening:
HPV testing
Multiple cervical cancer screening modalities are approved within existing cervical cancer screening guidelines. In developed countries, HPV testing has been approved for use in the UK and Australia. One test is already cleared in the US. In my opinion, the coming years will see a “sea change” away from cytology moving almost all of our screening to molecular tests of one sort or another. HPV testing is the poster child for this molecular focus. However, if employed at a scale, given that HPV testing has been shown to have only moderate specificity, it is evident that another secondary or intermediate test needs to be developed and implemented to avoid unnecessary, invasive, uncomfortable, and costly procedures. This secondary test needs to optimize specificity, without sacrificing sensitivity.
It is generally recognized that in one form or another, PCR testing for HPV is our current reference standard for this approach. Looking more closely at the technology, comparing the cycle threshold times (times it takes to return a positive result on the PCR test) suggests that higher-grade lesions return a positive result sooner than lower-grade lesions.
There is not yet a scalable solution based on this methodology but it presents an interesting area for further investigation of a secondary test that would prevent low-risk patients from undergoing more invasive testing, and yet would not involve additional procedures or visits.
Menstrual blood
Working with colleagues at Stanford University and Qvin, we have been investigating the value of menstrual blood for diagnostic and therapeutic purposes. As part of this effort we developed a special sanitary napkin, embedded with a test strip that can be removed and sent to the lab for evaluation. A number of tests can be run using the dried menstrual blood collected on the test strip, including FSH, cholesterol, HDL, Hemoglobin A1c, and a number of other everyday tests. That’s right, menstrual blood is, who would have thunk it, blood.
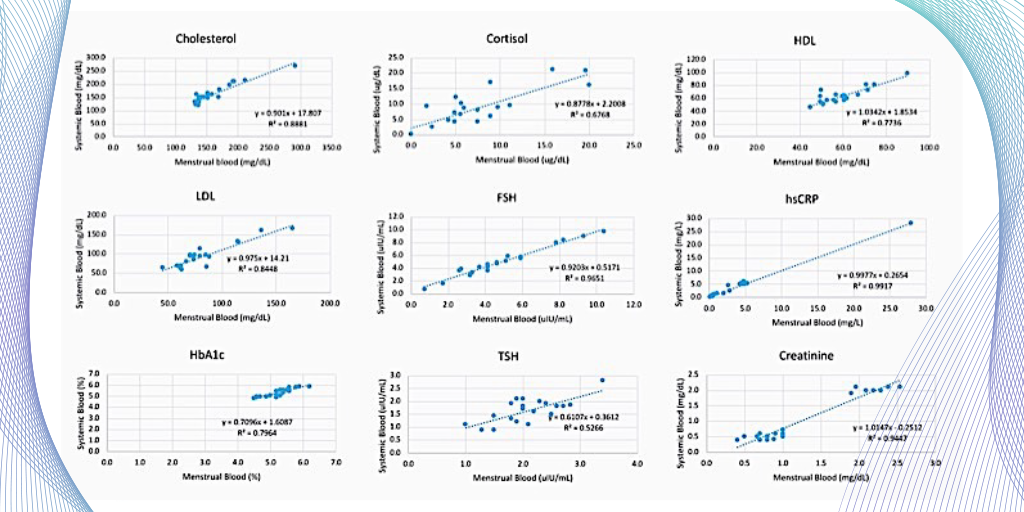
In addition, we just completed a study looking at HPV testing using this same menstrual pad, the QPad®. We were excited to find a 94-100% correlation in HPV from menstrual blood versus common HPV tests. The QPad also appeared to be more accurate and more acceptable to women than a self-swab of the vagina/cervix to obtain a specimen. Similar results have also been recently reported from China, so testing for HPV by menstrual blood without the need for an office visit or a clinical exam might be a possibility in the future.
Advances in colposcopy
VisualCheck AI computer-aided diagnostic
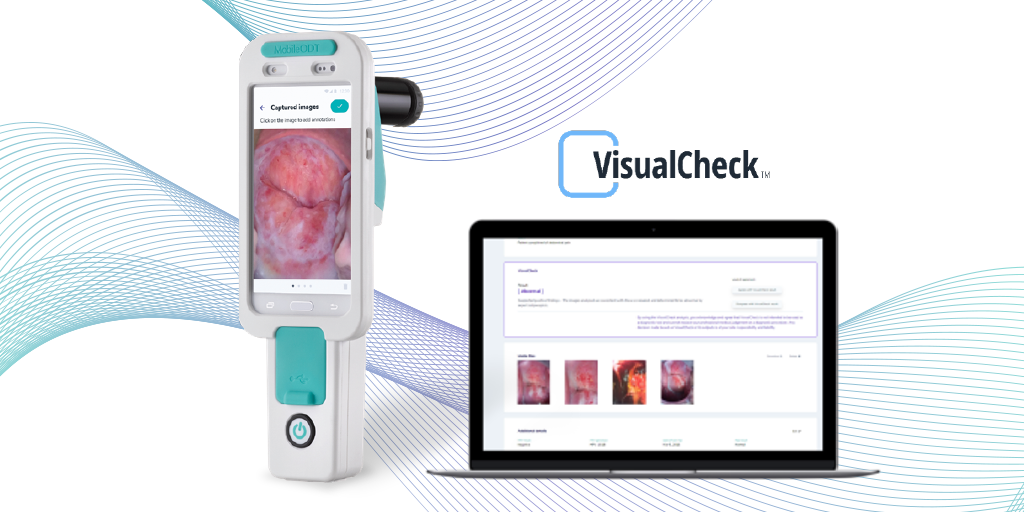
While VIA made a global impact on cervical cancer screening 20 years ago, the next ‘big thing’ in the realm of visual cervical assessment is VisualCheck, an AI-based, computer-aided diagnostic tool. VisualCheck runs on the EVA System digital colposcope and evaluates the risk of dysplasia at the point of care. VisualCheck compares the images captured in one exam with both thousands of previous exams annotated by leading experts and histology results, predicting how they would classify the images. The algorithm is calculated to recognize and identify what would be CIN2+ based on data obtained in 17 different countries.
Clinical evaluation of VisualCheck shows consistent agreement rates between the VisualCheck scores and expert panels in three separate geographies. VisualCheck is the only such computer-aided diagnostic tool that actually evaluates the likelihood of cervical dysplasia, offering hope to women without direct access to specialists.
Improvements in colposcopes
Besides hosting the VisualCheck algorithm, the EVA System digital colposcope offers a number of other advanced features to aid colposcopy. Completely portable and using Wi-Fi, EVA System enables immediate consultation, either elsewhere in the hospital or elsewhere on the planet, while captured images can be uploaded and shared, remotely. These images can be annotated, allowing for picture precise consultation, by specialists in other locations. In addition to images from the EVA, the system can be easily uploaded into the medical record providing both a visual record of a given exam and a historical record that can be useful to clinicians seeing the same patient in the future.
The consulting physicians are able to see where and when the biopsy was performed in real-time. For practitioners practicing in remote areas, or in areas with limited resources, this can mean the difference between a quick diagnosis and treatment or a prolonged, delayed course between testing and treatment.
Images produced on the EVA System are self-illuminating, enabling clearer image capture. Image assessment includes a green filter used to assess abnormal vessels and other landmarks or lesions. With autofocus, every image is assessed for a variety of factors: the presence of the cervix, illumination, the position of the cervix, and the focus of the image. Image-guided technology and machine-based learning allow the image to be assessed for image quality, notifying the clinician that the image is of suitable quality for actual visual inspection or colposcopic evaluation.
Advances in treatments
While thermal ablation is not new, it is still uncommon in the US. It’s an ablative approach similar to cryotherapy, but instead of freezing the cervical cells, it’s ‘heating’ them using the temperature at which water boil and this denaturing the cells in contact with the probe. Even a “double treatment” (two applications of the thermal probe) takes about a minute. The side effects are similar to cryotherapy with the advantage of having a 25mm probe for treatment of lesions on a larger, parous cervix for example, without the “spread” seen with conventional cryotherapy.
A variety of investigators are exploring the use of topical agents, oral medications, and even “therapeutic vaccines” to destroy abnormal cervical cells. A recent study used simple trichloroacetic acid, which you might be more accustomed to using for external warts. They used this for the treatment of low and high-grade dysplasia. The results were very positive, interestingly the HPV disappeared with the use of the trichloroacetic acid, and only one treatment was required. There will, no doubt, be a number of other studies focusing on such approaches in the future, again with the aim of simplifying treatment and making it more accessible to women globally.
Conclusion
There is little doubt that cervical cancer prevention paradigms will change radically in the coming years. Molecular technologies will be increasingly important as a primary screening approach. Alternative sampling approaches such as self swab and menstrual blood will replace conventional clinician obtained tests.
I expect to see reduced barriers to high-quality screening and treatment with better linkages between the two, and, as the use of AI-based approaches increases, we will enable expert-level cervical assessment anywhere in the world. I, for one, look forward to this new future.







